#Iso 9001:2015 vs iso 13485:2016 matrix iso
ISO 13485:2016 – How are they similar?īesides these differences in the structure, there are also similarities between ISO 9001:2015 and ISO 13485:2016: The ISO organization decided to release 13485 with the old structure.įor information on the differences between ISO 9001:2015 and ISO 9001:2008, see Infographic: ISO 9001:2015 vs. So, when ISO 9001:2015 was finally released with the new structure, 13485:2016 was also already ready for release with the ISO 9001:2008 structure. The new version of ISO 13485 was ready to be released in 2016 although it relied on ISO 9001:2008.
Unfortunately, corrections on the ISO 13485:2003 took a long time. The question everyone is asking is why 13485:2016 is so different from 9001:2015, when 13485:2016 came out just 6 months after 9001:2015 was released.

However, today the situation is slightly different because ISO 13485:2016 still supports the structure of old ISO 9001 revision (from 2008). Until the introduction of the high-level structure in 2015, ISO 9001 and ISO 13485 were very similar standards and it was very easy to build a quality management system that met the requirements of both of them.
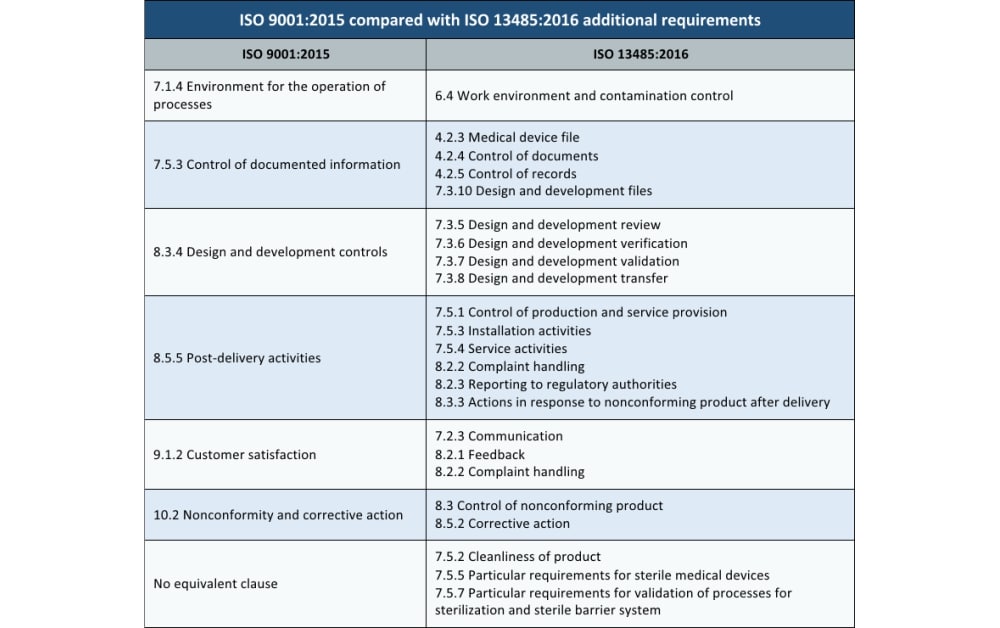
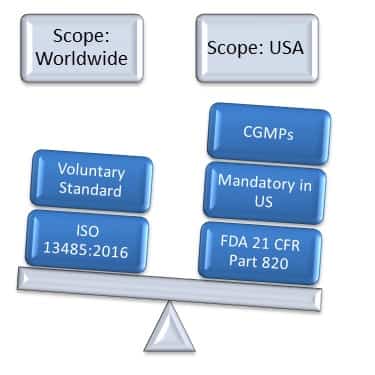
One major distinction of ISO 13485 is that it is intended to also be required for regulatory purposes as well as a non-statutory requirement for a quality management system. Just like these other standards, ISO 13485 includes the entire ISO 9001 standard with additional requirements included in blue italics text. Like many other quality management system requirements for special purposes (such as IATF 16949 for automotive production and service parts and AS9100 for use by aviation, space and defense organizations), the ISO 13485 standard is based on the requirements of ISO 9001. ISO 13485 is the international standard requirement for a medical device quality management system. Note: This article was updated according to the ISO 13485:2016 revision.
